Today we learned about Genghis Khan. We wanted to do an arts and crafts project, but let’s face it Genghis Khan wasn’t an ‘artsy craftsy’ fellow so, we decided to work off of the Chinese portion of the lesson and make some Chinese Lanterns.
I was surprised by the excitement of my children toward this simple craft! So I wanted to share this lesson with you.
Materials: (makes one lantern)
1, 11″x18″ piece of construction paper
1 Chop stick
2 pieces of string (approximately 18″ each)
Masking Tape
1 Flameless tea light
Preparing the paper:
First we took a standard 11″x18″ piece of construction paper and folded it in half ‘hot dog’ style.
Then we measured with our rulers and marked off 1″ increments along the long edge of the paper.
We made an additional line at the opposite side of our markings along the entire edge about 1″ in from the edge. (This would be the place to stop cutting.)
Finally we cut straight lines from our measured markings to the ‘stop line’.
Assembly:
We opened up the folded paper and curled the opened paper into the lantern shape. Then stapled the two short ends of the construction paper together at the top and the bottom of the lantern.
We cut one small strip of paper and fastened it across the bottom of the lantern to hold our candles. (flameless tea lights)
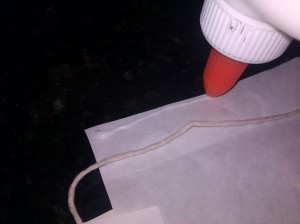
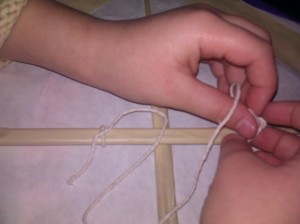
We stapled the strings to the top of our lantern and attached them to a stick with tape.
That’s it. My children took their finished lanterns into a darkened room and bounced them up and down. They both remarked how much the finished lanterns looked like ‘jellyfish’ with the tea lights reflecting inside.
With Chinese New Year coming up I thought this activity also would be fun to share with some of our book club friends too. I hope your children enjoy this craft as much as mine did.