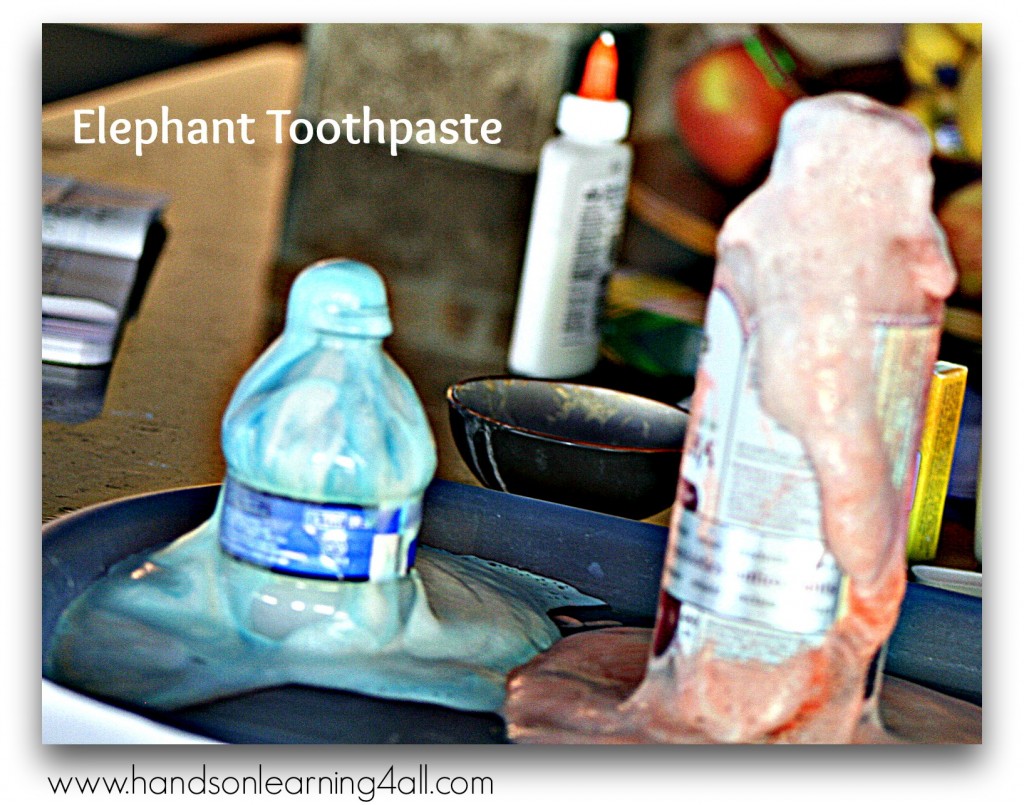
Elephant Toothpaste has been floating around the internet long enough for the Mythbusters to take notice and do a segment on it. (http://www.youtube.com/watch?v=FvGJ3LZbhDA&list=PL0A5590CEE7F2EC3B&index=3 Skip ahead to 1:55)
So what is this stuff? Is it really toothpaste for elephants? No…but this experiment does have a VERY cool reaction and can be used to demonstrate an exothermic reaction. Here’s the definition for an exothermic reaction: An exothermic reaction is a chemical reaction that releases energy in the form of light or heat. This experiment emits HEAT! Make sure that you exercise safety when pouring the peroxide into the bottle and with the chemical results while the experiment is occurring. After the chemicals have reacted completely the foam is just water, soap and oxygen so it is safe to touch and clean up by rinsing down the drain. YOU DEFINITELY DON’T WANT TO EAT THE FOAM! (Just in case you get any ideas of using the foam as real toothpaste….DON’T!)
Here’s the ingredient list:
•1 clean 16 ounce plastic/glass bottle
•1 clean pint sized water bottle (the smallest one you can find)
•1 tray
•1/2 cup 40-volume Hydrogen Peroxide Liquid (40-volume is a 12.12% solution, ask an adult to get this from a beauty supply store or hair salon)
•1 One Packet of Dry Yeast (about 1 1/4 tablespoons)
•3 Tablespoons of Warm Water
•1 Tablespoon of Liquid Dish Washing Soap
•10 drops of Food Coloring (your choice of color)
•Small cup
•Plastic Gloves
•Safety Goggles/Glasses
What to do:
***Place the bottle onto the tray–>this will help you clean up once your ‘elephant toothpaste’ has erupted!***
1. In the small cup mix the warm water with the yeast packet and set it aside for 30 second to 1 min.
2. Have an adult put on the gloves and the safety goggles. Measure and pour the hydrogen peroxide into the bottle. (Remember the bottle should be on the tray.)
3. Add the food coloring to the bottle.
4. Add the dish soap to the bottle and gently agitate the bottle to combine your ingredients.
5. Finally pour in the yeast/water solution. Do this quickly because your reaction will occur as a result of this step.
Take notes! The foam will begin to rise inside the bottle. You will begin to feel heat coming from the foam once it begins to flow from the bottle. The heat is a result of your chemical reaction and it is called an exothermic reaction.
Now formulate a hypothesis: What will happen if you use the smaller bottle for the same experiment. Try it. What happened? Were you right? Why or why not? Take pictures and write about your experiment!
My kids LOVED this experiment. If you can’t get the salon hydrogen peroxide, regular grocery store peroxide (for minor cuts, etc.) will work…your reaction just won’t be as powerful. This is a wonderful opportunity to get your reluctant writers to write about what would happen if you changed the amounts of the ingredients or used a larger bottle instead of a smaller one. The possibilities are almost endless.